
PPT - Base Excess and Critical Care in the past and in the future PowerPoint Presentation - ID:4963280

Noncarbonic buffer power calculated according to formula used in the... | Download Scientific Diagram

Oxidation of hydroxide ions in weak basic solutions using boron-doped diamond electrodes: effect of the buffer capacity - Analyst (RSC Publishing) DOI:10.1039/C9AN00505F
THE STANDARD STRONG ION DIFFERENCE, STANDARD TOTAL TITRATABLE BASE, AND THEIR RELATIONSHIP TO THE BOSTON COMPENSATION RULES AND
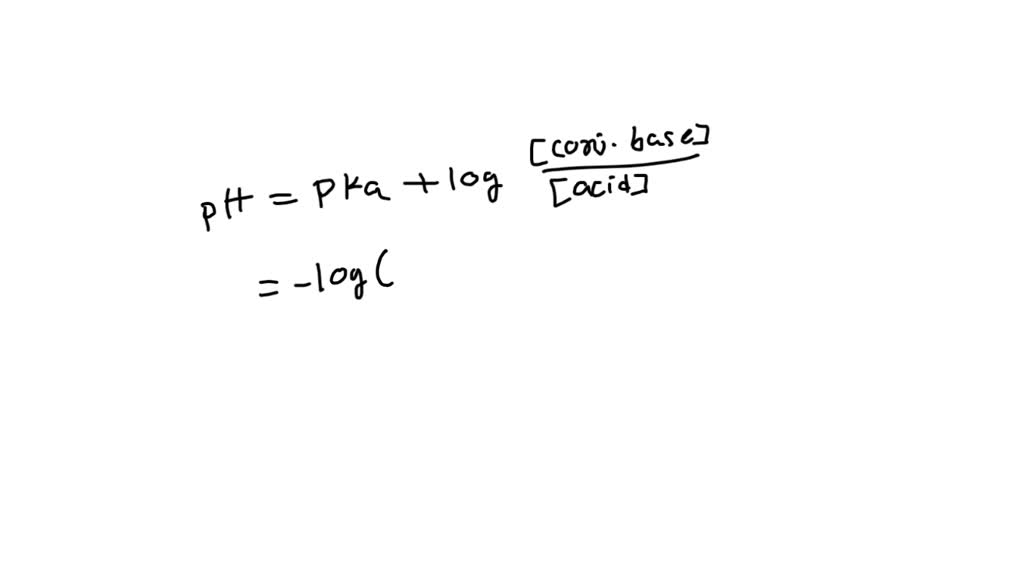
VIDEO solution: Hi I hope you can help me with this 3. Give the Van Slyke' Equation for: 3.1 Weak electrolytes and their salt 3.2 Strong acid solutions as buffer (applicable if
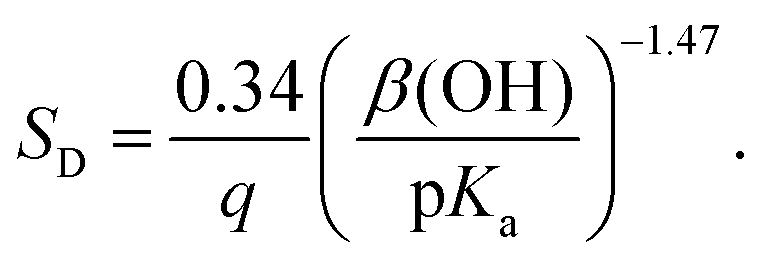
Energy and techno-economic analysis of bio-based carboxylic acid recovery by adsorption - Green Chemistry (RSC Publishing) DOI:10.1039/D1GC01002F
















