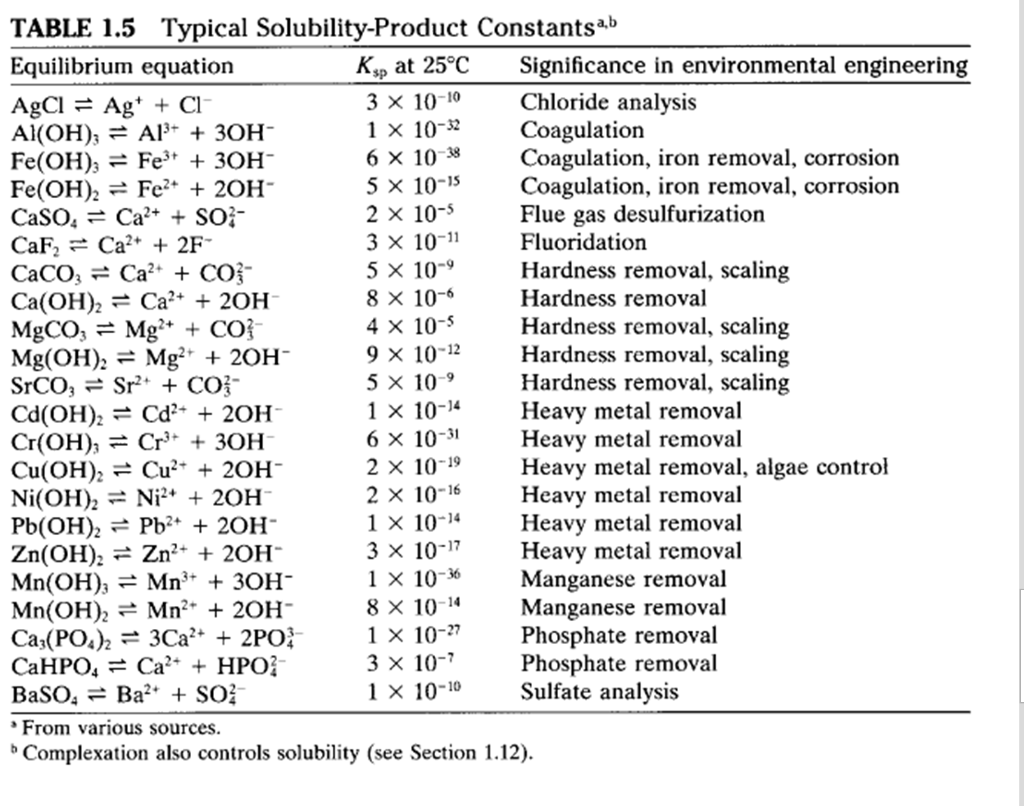
SOLVED: TABLE 17.2 Selected Solubility-Product Constants (Ksp) at 25*C Compound Formula Kap Compound Barium fluoride BaFz 2.45 * 10- 5 Leadllll chloride Formula Ksp 1,.17 * 10 PbClz Barium sulfate BaSO4 1,07
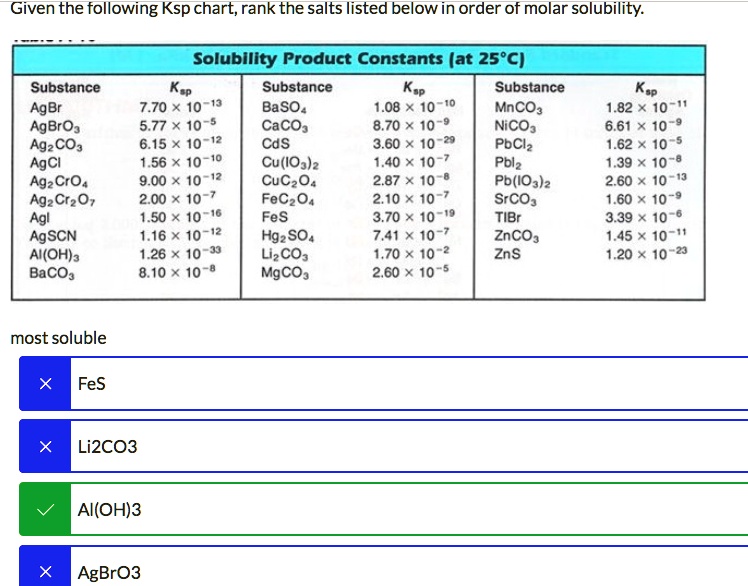
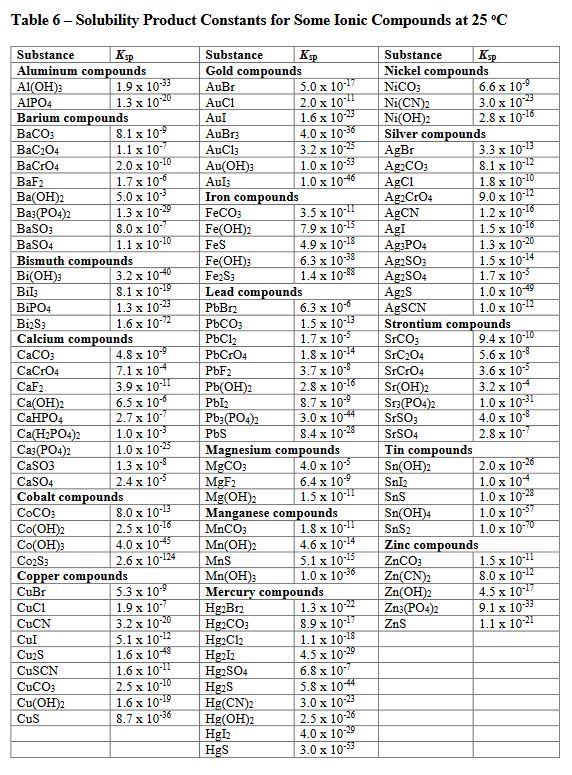
SOLVED: Solubility Product Constants (at 25°C) Ksp Substance Ksp Substance 7.70 * 10^-13 BaSO4 1.08 MnCO3 5.77 * 10^-5 CaCO3 8.70 NiCO3 6.15 * 10^-6 CdS 3.60 PbCl2 1.56 * 10^-10 Cu(IO3)2
![Table 1 from Dissolution and Solubility Product of Cd-Fluorapatite [Cd5(PO4)3F] at pH of 2–9 and 25–45°C | Semantic Scholar Table 1 from Dissolution and Solubility Product of Cd-Fluorapatite [Cd5(PO4)3F] at pH of 2–9 and 25–45°C | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/06aa56ea43681c5ca71879d7ff5d8c651f666f6c/8-Table1-1.png)
Table 1 from Dissolution and Solubility Product of Cd-Fluorapatite [Cd5(PO4)3F] at pH of 2–9 and 25–45°C | Semantic Scholar
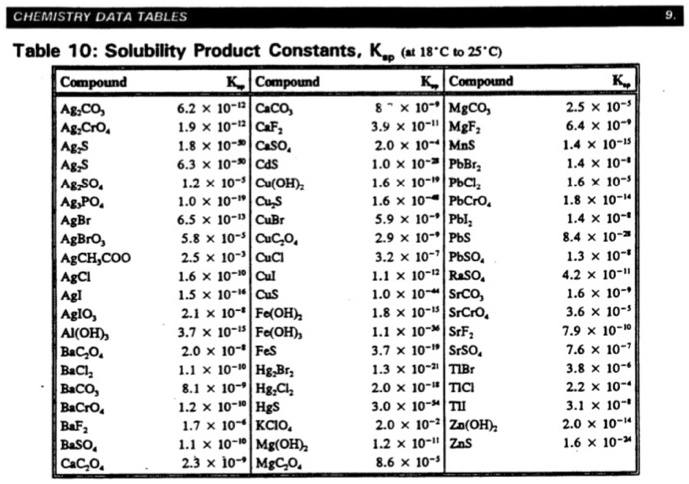
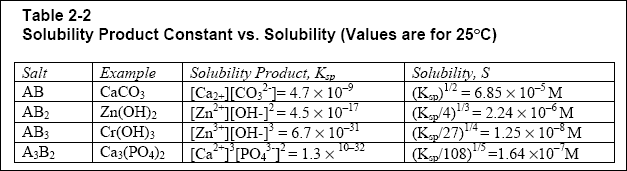
SOLVED: Table 10: Solubility Product Constants, K (18°C to 25°C) Compound K ABCO 6.2 * 10^-8 MgCO3 1.9 * 10^-11 CF2 3.9 * 10^-12 MgF2 1.8 * 10^-10 CSO2 2.0 * 10^-9

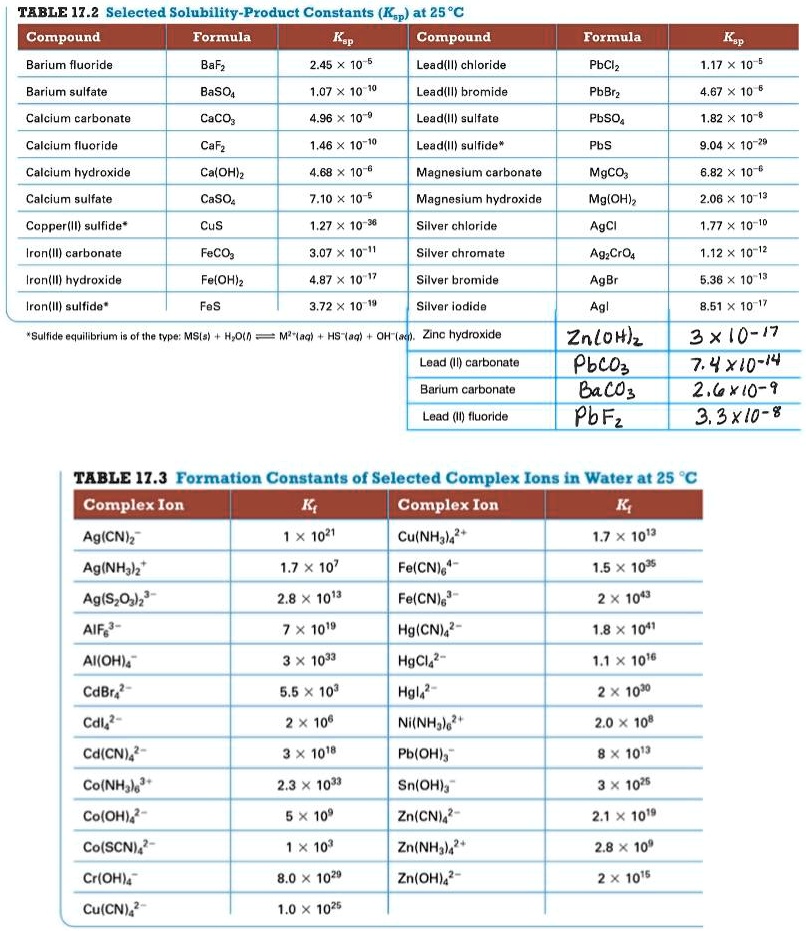
Table of Ksp values.pdf - Table of Solubility Products Compound Aluminium Hydroxide anhydrous Formula Al OH 3 Temperature 20C Ksp 1.91033 34 Aluminium | Course Hero